- Blog
- 3dsimed files
- Dam japanese karaoke list
- Grand theft auto 2 wii u
- Snapgene viewer change color
- Remington gun safes at costco
- Download do daemon tools gratis
- Index of ant man and the wasp hindi
- Jogos de pes 2010
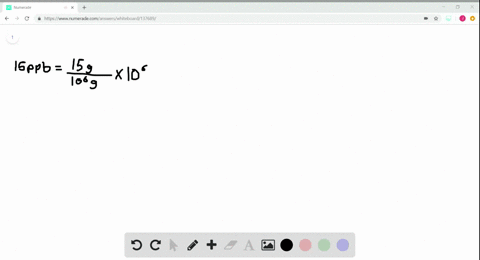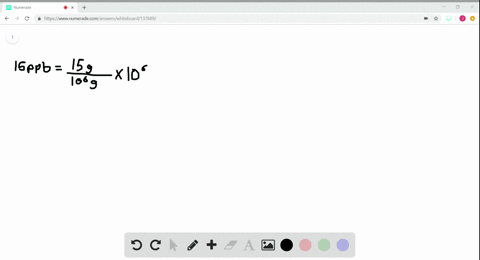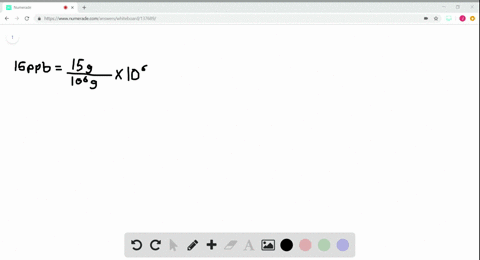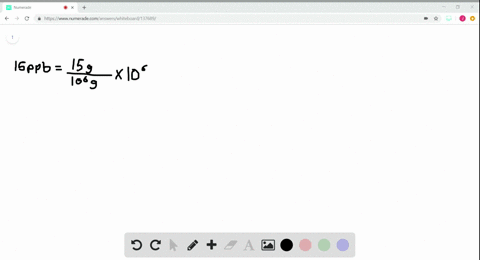
- How to calculate ppm and ppb
- Crow zero film complet vf
- How to improve company size wmma 5
- Swords and sandals 4 online game
- Blog
- 3dsimed files
- Dam japanese karaoke list
- Grand theft auto 2 wii u
- Snapgene viewer change color
- Remington gun safes at costco
- Download do daemon tools gratis
- Index of ant man and the wasp hindi
- Jogos de pes 2010
- How to calculate ppm and ppb
- Crow zero film complet vf
- How to improve company size wmma 5
- Swords and sandals 4 online game
How will you prepare 100 ppm of ammonia solution?Īmmonia Standard, 100 ppm NH3 as N. To avoid waste, and depending on the volumes you need, you can proportionally cut the volume of all your stocks. Then, re-run the calculation using the dilution formula. Take 0.1 mL of your 1000 ppm stock and dilute it to 100 mL (use a volumetric flask for accuracy and convenience). The 200 ppm NaOCl means: there are 200 g of NaOCl in every 1000000 g of dilute solution. Zinc Standard Solution (100 ppm Zn): Dissolve 0.440 g of zinc sulfate in water containing 1 ml of 5M acetic acid and add sufficient water to produce 100.0 ml. Zinc Standard Solution (25 ppm Zn): Dilute 25 volumes of zinc standard solution (100 ppm Zn) to 100 volumes with water. PPM is derived from the fact that the density of water is taken as 1kg/L = 1,000,000 mg/L, and 1mg/L is 1mg/1,000,000mg or one part in one million. One thousanth of a gram is one milligram and 1000 ml is one liter, so that 1 ppm = 1 mg per liter = mg/Liter. Convert the mass in grams to a mass in milligrams: mass NaCl = 0.0045 g = 0.0045 g × 1000 mg/g = 4.5 mg.Extract the data from the question: mass solute (NaCl) = 0.0045 g.Write an equation representing the ppm concentration: ppm = mass solute (mg) ÷ volume solution (L).Parts Per Million (ppm) Concentration Calculations To convert to parts per million, you don’t need to do anything other than take the mg/kg off the end and add ppm on to the end. Why am I having to backwash my pool so often?Īssuming you are converting the mass of a substance, the conversion of mg/kg to ppm is 1:1.How many hours a day should a swimming pool pump run?.

How long should you backwash a pool for?.How do you tell if there is too much chlorine in a pool?.